How Biotech Secrets Shape FDA Risks and Safeguards?
Analysis reveals 9 key thematic connections.
Key Findings
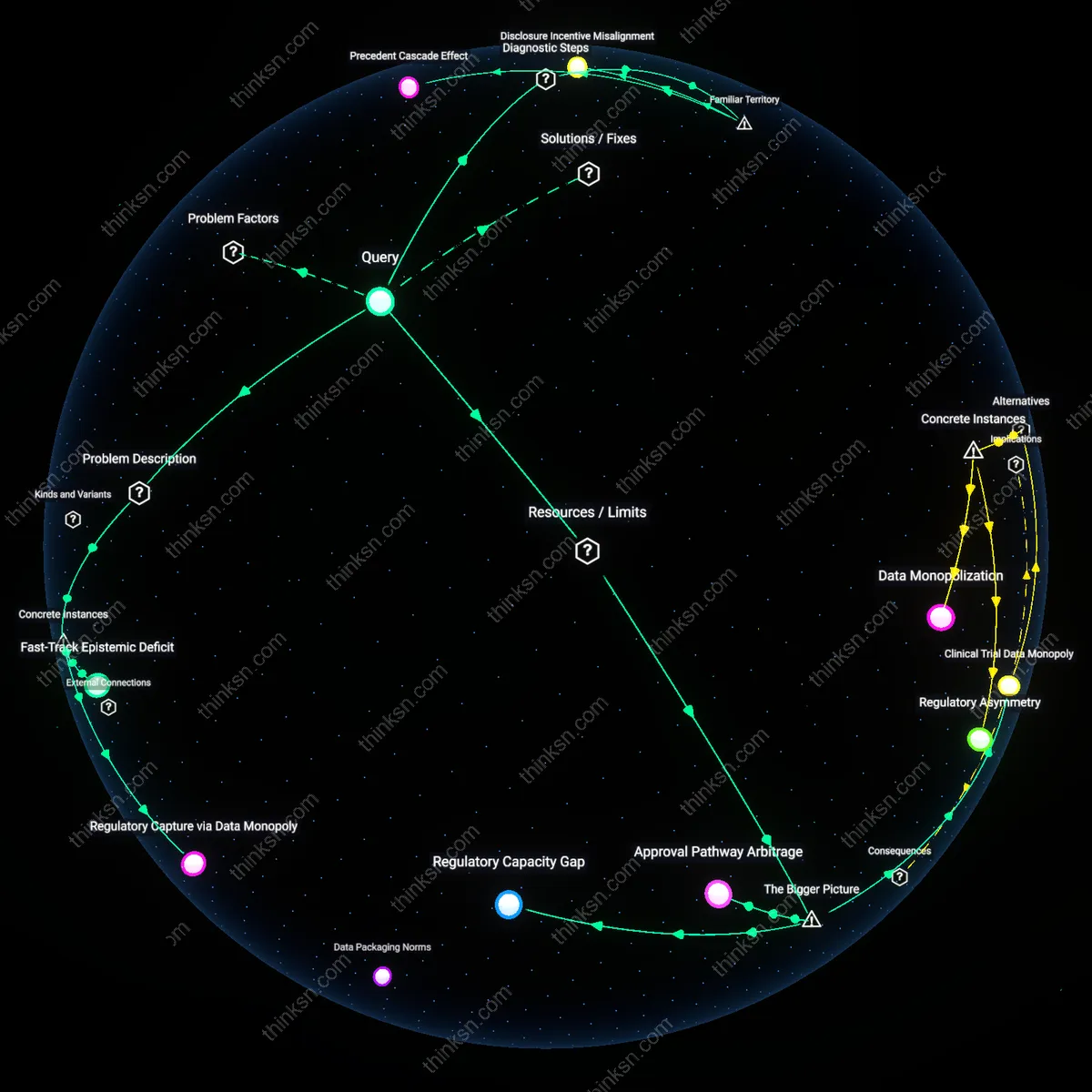
Regulatory Capture via Data Monopoly
Biotech firms uniquely control pre-approval clinical trial data, preventing the FDA from independently verifying safety signals, as seen in the 2004 Vioxx (rofecoxib) withdrawal where Merck delayed disclosing cardiovascular risk data from the APPROVe trial; this asymmetry allows firms to frame risk assessments around selective endpoint reporting, and the FDA’s reliance on submitted datasets—rather than adversarial validation—enables temporal concealment of adverse events, a dynamic underappreciated because regulatory review is perceived as scientifically autonomous when in fact it is epistemically dependent.
Fast-Track Epistemic Deficit
The FDA’s Accelerated Approval pathway for oncology drugs, exemplified by the 2021 approval of Aduhelm (aducanumab) despite strong advisory committee dissent, reflects how information asymmetry skews risk assessment when firms like Biogen submit complex, narrowly interpreted biomarker data that only they fully understand; the agency’s lack of access to raw, real-time trial data and its dependence on sponsor-analyzed results creates a cognitive lag, where decision-makers are forced to assess risks based on narrative summaries rather than primary evidence, a vulnerability that is structurally embedded but rarely acknowledged in regulatory timelines.
Post-Market Surveillance Arbitrage
When Vertex Pharmaceuticals submitted Phase 3 data for Trikafta (elexacaftor/tezacaftor/ivacaftor) in 2019, the FDA approved the therapy for cystic fibrosis with limited long-term safety data, relying on surrogate endpoints while deferring robust risk detection to post-market monitoring—a shift that exploits asymmetric information by transferring the burden of evidence generation to public health systems; this creates a blind spot where rare but severe hepatic adverse events only emerged after widespread use, revealing that the practical safeguard of mandatory post-approval studies (REMS) is routinely underenforced due to regulatory resource asymmetry, a dynamic obscured by the rhetoric of 'patient access.'
Regulatory Capture Risk
FDA advisory committees' reliance on industry-funded clinical trial data distorts risk assessment by embedding biotech firms' selective disclosures into official evaluation frameworks. Biotech sponsors control the timing, format, and completeness of data submitted during New Drug Application reviews, and because FDA reviewers depend on these submissions to assess safety and efficacy, gaps or framing biases in the evidence directly shape regulatory conclusions. The mechanism operates through the FDA’s resource-constrained review model, which cannot independently replicate trials, making it structurally vulnerable to asymmetrical access. The underappreciated reality is that transparency is not just about withholding data—it’s about firms strategically constructing the evidentiary landscape, which feels familiar because the public associates FDA trust with data integrity, not data provenance.
Precedent Cascade Effect
The FDA’s tendency to align future risk benchmarks with prior approvals from asymmetric information environments creates self-reinforcing standards that accommodate incomplete disclosure. When reviewers approve a drug based on limited or sponsored data, subsequent applications reference that decision as justification for similar data packages, propagating the initial information imbalance across drug classes and therapeutic areas. This operates through the formal reliance on precedent in regulatory decision-making, particularly in fast-tracked or orphan drug pathways, where historical leniency becomes institutionalized. The non-obvious insight is that even when new data emerges, the system resists recalibration because deviation from precedent appears riskier than perpetuating inherited gaps—mirroring public assumptions that regulatory consistency equals safety, when it may instead entrench asymmetry.
Disclosure Incentive Misalignment
Biotech firms face weaker internal incentives to report negative trial outcomes than to emphasize efficacy signals, and this behavioral asymmetry directly infiltrates FDA risk models that assume balanced reporting. Because financial valuation and investor attention reward positive milestones, companies systematically delay, minimize, or reframe adverse findings in submissions, while the FDA lacks real-time monitoring tools to detect such omissions. This dynamic plays out in the ClinicalTrials.gov reporting lag and selective subgroup analyses, where omissions are not illegal per se but distort cumulative risk interpretation. The overlooked aspect is that the system assumes strategic withholding is rare, even though market logic makes it predictable—echoing the widespread but unexamined belief that regulatory filing equals full transparency.
Regulatory Capacity Gap
The FDA's risk assessment is systematically weakened when biotech firms possess proprietary biomarker algorithms that are not subject to external validation, because the agency lacks both the statutory authority and technical infrastructure to audit AI-driven drug discovery models in real time. This creates a Regulatory Capacity Gap, where the FDA’s reliance on submitted trial outcomes — rather than access to raw computational logic — insulates flawed assumptions from detection, especially in personalized oncology therapies. Crucially, this imbalance persists not due to regulatory indifference but because digital tool validation remains outside the current CDER review templates, making it an institutional blind spot rather than a solvable data gap.
Clinical Trial Data Monopoly
Sponsors’ exclusive control over early-phase trial datasets distorts the FDA’s risk calibration by allowing selective reporting of efficacy signals while delaying disclosure of adverse event clusters until post-marketing phases. Because the FDA cannot mandate shared access to real-time safety dashboards during Phase II, firms can exploit staggered reporting norms to present risk profiles that appear favorable at approval milestones. This dynamic is amplified in rare disease domains — such as gene therapies for spinal muscular atrophy — where small cohorts make outlier suppression consequential, revealing a systemically enabled obfuscation mechanism rooted in data ownership asymmetry rather than outright deception.
Approval Pathway Arbitrage
Biotech firms manipulate information asymmetry by strategically routing high-uncertainty products through expedited pathways like Breakthrough Therapy Designation, where evidentiary thresholds are lower and review timelines compressed, thereby curtailing the FDA’s capacity to commission independent modeling or long-term risk simulations. This arbitrage is structurally enabled by congressional mandates to accelerate access, which shift the burden of proof onto post-marketing surveillance systems like Sentinel — systems that are underfunded and reactive, not preventive. The result is a feedback loop where speed concessions become permanent entry points for data-poor approvals, embedding systemic risk tolerance into regulatory culture.