Does FDA Clearance Mean Trust When Judges Are Former Execs?
Analysis reveals 11 key thematic connections.
Key Findings
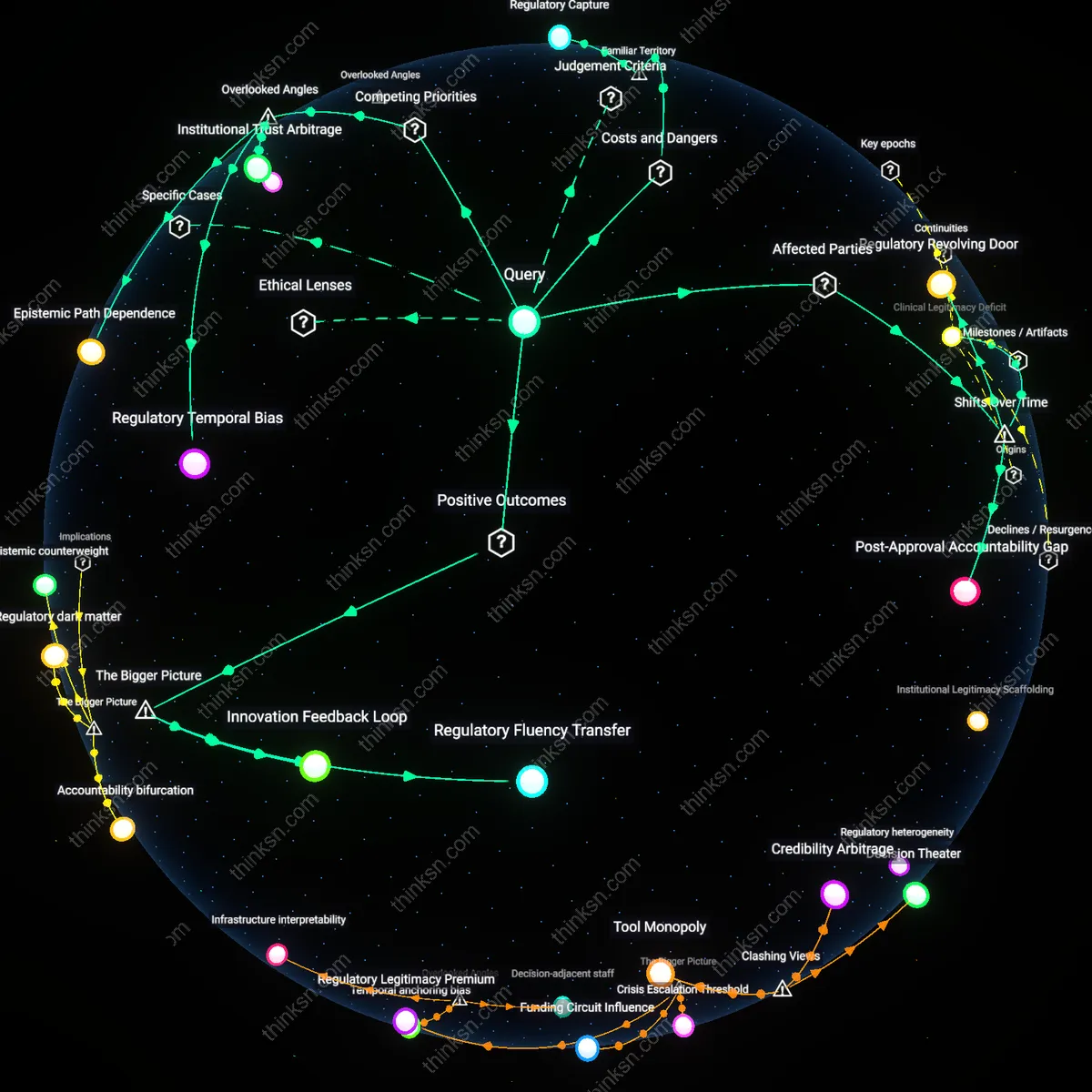
Regulatory Revolving Door
Consumers should interpret FDA clearance as potentially compromised when approval panels include former manufacturer executives because the post-1997 shift toward industry-funded user fees under the FDA Modernization Act created structural incentives for staffing overlap. As the FDA increasingly relied on device makers to fund review processes, former industry personnel were hired into advisory and review roles, normalizing a career trajectory between regulatory service and corporate employment. This institutionalized mobility—once rare before the 1990s—transforms the interpretation of clearance from independent validation to a signal of negotiated acceptability, revealing how financial dependency reshaped regulatory epistemology.
Clinical Legitimacy Deficit
Consumers should interpret FDA clearance skeptically when former executives sit on approval panels because the early 2000s expansion of the 510(k) pathway outsourced safety validation to historical equivalence, weakening clinical scrutiny just as industry migration into regulatory roles rose. As panel decisions began relying on devices already deemed safe—often developed by the same firms employing review alumni—the presumption of equivalence masked cumulative risk, and the interpretive authority of clearance shifted from evidence of innovation to affirmation of market continuity. This dynamic, crystallized after the 2007 reauthorization of user fees, reveals a legitimacy deficit where regulatory trust is no longer anchored in trial data but in institutional familiarity.
Post-Approval Accountability Gap
Consumers should interpret FDA clearance as an increasingly provisional judgment because the 2012 creation of the Breakthrough Devices Program accelerated pre-market approval while shifting evidentiary burdens to post-market surveillance, coinciding with the integration of industry-experienced personnel into fast-track panels. With former executives facilitating expedited reviews under time-bound mandates, the clearance event itself becomes less a conclusion than a starting point for data collection, yet public perception lags behind this procedural shift. The gap between provisional regulatory approval and consumer belief in definitive safety—amplified since 2016 in high-profile device recalls—exposes an accountability gap where responsibility dissolves across time and institutional phases.
Regulatory Fluency Transfer
FDA clearance gains efficiency when former manufacturer executives join review panels because their insider knowledge of device development timelines, manufacturing constraints, and clinical integration pathways accelerates alignment between regulatory expectations and real-world implementation; this transfer of operational fluency reduces redundant data requests and miscommunication, enabling faster market entry without compromising safety benchmarks—what appears as conflict of interest may in fact activate a hidden conduit of institutional learning. The mechanism hinges on former executives internalizing both corporate execution logic and regulatory standards, allowing them to anticipate evidentiary gaps before submission, thus compressing review cycles through predictive compliance rather than preferential treatment.
Innovation Feedback Loop
The presence of former manufacturer executives on FDA panels strengthens a feedback loop between post-market performance data and pre-market design refinement, as these individuals carry tacit knowledge about how devices fail in clinical settings back into the evaluation phase of new iterations; this creates a system where regulatory scrutiny becomes more clinically grounded and responsive to actual use patterns. Hospitals, insurers, and patients benefit because cleared devices are not only compliant but increasingly calibrated to real-world usability and failure modes—this advantage emerges not from leniency but from the panel’s ability to simulate downstream integration challenges better than purely academic or bureaucratic reviewers.
Standards Co-Evolution
When former industry executives participate in FDA clearances, they help co-evolve technical and evidentiary standards by embedding emerging engineering paradigms—such as AI-driven validation or digital twin modeling—into acceptable regulatory frameworks, ensuring that clearance criteria keep pace with technological change rather than lag behind it; this dynamic enables the FDA to maintain legitimacy in fast-moving fields like connected medical devices or personalized diagnostics. The systemic value lies in preventing regulatory obsolescence, where outdated assessment methods stifle innovation or misclassify novel technologies as high-risk due to unfamiliarity rather than actual hazard.
Regulatory Capture
Consumers should treat FDA clearance as compromised when approval panels include former manufacturer executives because direct career mobility between industry and regulators creates decision-making bias. This mechanism—where individuals with financial or professional ties to a company participate in its oversight—enables preferential treatment through lenient review standards, especially in high-stakes, data-complex devices like implantables or diagnostics. The non-obvious risk is not outright fraud but the normalization of industry-aligned interpretations of safety and efficacy, which erodes the FDA’s role as an independent gatekeeper.
Trusted Agent Failure
Consumers should assume that FDA clearance in such cases reflects institutional betrayal rather than assurance because the presence of ex-executives transforms public oversight into a symbolic endorsement. This dynamic operates through the public’s reliance on the FDA as a trusted agent—a culturally ingrained reflex after decades of associating the agency with protection from harmful drugs and devices. The underappreciated danger is not corruption per se, but the collapse of trust when a guardian institution validates products while staffing its panels with those who previously marketed them.
Regulatory Temporal Bias
Consumers should interpret FDA clearance with skepticism when approval panels include former manufacturer executives because recurrent personnel movement compresses the agency’s evaluative timeline, privileging speed over longitudinal safety assessment. The revolving door between industry and regulation incentivizes executives-turned-advisors to expedite approvals using familiar, precedent-based metrics, implicitly devaluing long-term monitoring systems that lack immediate visibility. This bias toward near-term validation disrupts the temporal integrity of regulatory scrutiny—a dimension rarely highlighted, as most critiques focus on financial conflicts, not the erosion of time-bound diligence essential for detecting latent device risks.
Epistemic Path Dependence
Consumers should question the neutrality of FDA clearance in such cases because former manufacturer executives bring embedded cognitive frameworks that subtly shape evidentiary thresholds, favoring data types their prior firms optimized for. This path dependence entrenches specific validation paradigms—like short-duration clinical outcomes or surrogate endpoints—over more diverse or patient-centered measures, not through overt manipulation but through routine methodological choices in trial design and endpoint selection. The overlooked issue is that conflicts of interest operate cognitively, not just financially, altering what counts as ‘proof’ in ways that perpetuate legacy device evaluation models at the cost of methodological innovation.
Institutional Trust Arbitrage
Consumers should recognize that FDA clearance under these conditions functions as a strategic transfer of reputational capital, where manufacturers leverage the agency’s public trust to offset private liability risks. Former executives, fluent in both corporate and regulatory discourse, act as conduits who convert FDA endorsement into market legitimacy while insulating their former firms from full accountability—effectively arbitraging public confidence for private gain. This dynamic is rarely acknowledged because discourse centers on individual ethics, not the systemic repurposing of regulatory credibility as a fungible asset in post-approval commercial and legal strategy.