When PSA Screening Harms More Than Helps: The Overdiagnosis Threshold?
Analysis reveals 7 key thematic connections.
Key Findings
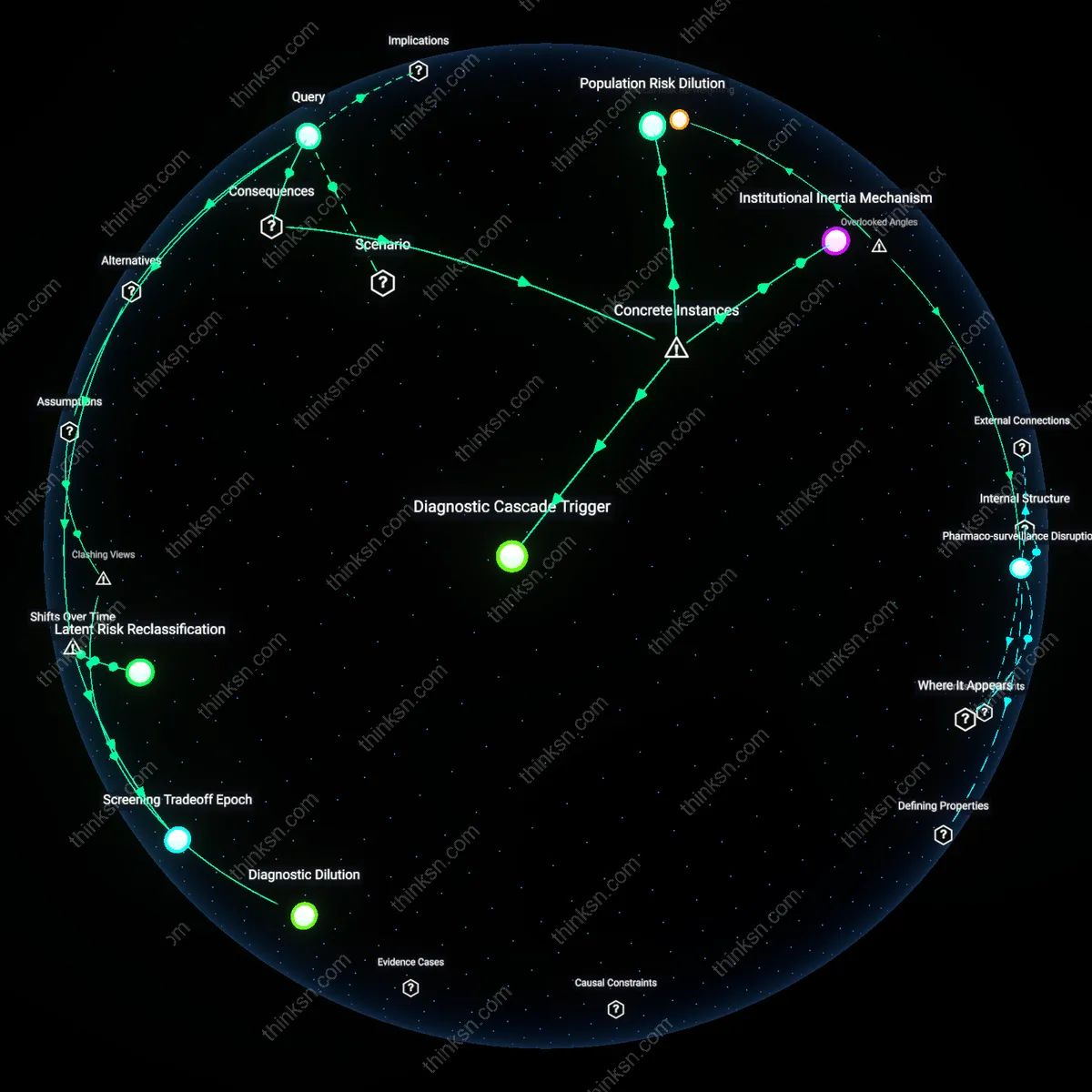
Diagnostic Dilution
Overdiagnosis exceeds benefit in PSA screening when more than 25% of detected prostate cancers represent indolent disease unlikely to cause morbidity, because population-level data from the European Randomized Study of Screening for Prostate Cancer (ERSPC) shows that 750 men must be screened to prevent one prostate cancer death, with over 50% of screen-detected tumors classified as low-risk on biopsy; this mechanism—whereby screening expands the pool of diagnosed cases while minimally affecting mortality—reveals that clinical benefit is eroded not by overtreatment alone, but by the progressive weakening of diagnostic specificity in large-scale screening systems, a non-obvious consequence of defining disease solely through biomarker thresholds rather than clinical behavior.
Latency Arbitrage
Routine PSA screening generates net harm when the mean overdiagnosis threshold falls below 40%, because actuarial models from the Surveillance, Epidemiology, and End Results (SEER) program demonstrate that average-risk men over 60 have a 10-year prostate cancer-specific mortality of less than 3%, while aggressive monitoring and treatment shift cause of death from cancer to cardiovascular or treatment-related complications; the dominant risk-benefit calculus assumes synchronous disease progression, but in reality, screening exploits temporal gaps between detection and clinical manifestation, allowing intervention to claim credit for indolent disease regression, thereby masking the fact that mortality reduction is achieved at the cost of systematically shortening the diagnostic timeline for non-lethal conditions.
Diagnostic Cascade Trigger
In the 1990s in the United States, widespread PSA screening led to a surge in prostate biopsies and subsequent overtreatment of low-risk tumors identified through the Veterans Health Administration and Medicare data, revealing that once a man receives an abnormal PSA result, the clinical pathway almost invariably leads to further invasive procedures regardless of actual risk, because the healthcare system incentivizes intervention over surveillance and clinicians fear malpractice liability for under-treatment; this cascade converts indolent disease into active management, making overdiagnosis functionally irreversible and amplifying downstream harms well beyond the initial false positive, thereby establishing that even modest overdiagnosis rates become harmful when embedded in a system structured to escalate care.
Population Risk Dilution
In the European Randomized Study of Screening for Prostate Cancer (ERSPC), site-specific variation—particularly between the Rotterdam arm and others—demonstrated that screening yields greater net benefit in concentrated, high-incidence populations with targeted uptake, whereas when PSA testing was generalized across broader, average-risk Dutch cohorts, the proportion of men diagnosed with clinically insignificant cancer rose sharply, diluting the mortality benefit across a larger pool of low-risk individuals and exposing more men to treatment complications without survival gain, revealing that the threshold for harm exceeds benefit not due to test inaccuracy alone but because public health deployment in heterogeneous populations weakens risk stratification, a dynamic overlooked when evaluating trials with selective enrollment.
Institutional Inertia Mechanism
After the 2012 US Preventive Services Task Force (USPSTF) recommendation against routine PSA screening, uptake declined only marginally in community urology practices in states like Florida and Texas, where fee-for-service models and patient demand sustained high screening volumes despite evidence of net harm at population levels, showing that once screening becomes embedded in clinical norms and revenue streams, the threshold for overdiagnosis that tips the balance toward harm is effectively raised by organizational resistance to de-implementation, meaning that the real tipping point is not epidemiological but institutional—where systemic momentum outweighs scientific recalibration.
Screening Tradeoff Epoch
The threshold prevalence of overdiagnosis at which PSA screening causes net harm shifted decisively after the 2008 U.S. Preventive Services Task Force draft recommendation, when epidemiological evidence from randomized trials like ERSPC and PLCO was reinterpreted through a time-bounded calculus of cumulative morbidity. Urologists, epidemiologists, and policy panels began weighing the delayed benefits of reduced prostate cancer mortality against the immediate and accumulating harms of false positives and overtreatment, revealing that once overdiagnosis exceeded 25–30% of detected cases, population-level harm outweighed benefit. This recalibration was not evident before the mid-2000s, when screening assumed latent cancers were uniformly progressive; the shift exposed a new temporal logic in preventive medicine—the point at which prolonged observation overtakes intervention as the ethical default.
Latent Risk Reclassification
Overdiagnosis crossed the harm-benefit threshold in 1992–1995, when serum PSA testing became routinized in primary care clinics across North America, transforming the epidemiological profile of prostate cancer from a clinically apparent disease to one dominated by subclinical phenotypes. As family physicians, rather than urologists, became the primary referrers, the diagnostic cascade accelerated, detecting a much larger reservoir of indolent tumors that would not have progressed within a man’s lifetime. The significance lies not in the eventual mortality data—which came later—but in this earlier systemic shift in diagnostic authority and network flow, which silently reclassified entire cohorts of asymptomatic men as 'at risk,' making overdiagnosis structurally inevitable before its magnitude could be measured.