Do Neuromodulation Devices Outweigh Surgery Risk with Fewer Migraines?
Analysis reveals 10 key thematic connections.
Key Findings
Therapeutic Citizenship
Chronic migraine patients should evaluate surgical trade-offs by recognizing their evolving role as co-managers of a biomedical identity shaped by post-2000s patient advocacy and device industry collaboration. As neuromodulation transitioned from experimental to clinically sanctioned in the 2010s, patients became enrolled in a new therapeutic class—licensed to access devices like occipital nerve stimulators—not solely through medical criteria but through their capacity to navigate insurance approvals, comply with device monitoring, and report outcomes in ways that validate long-term cost-effectiveness; this shift redefined patient agency not as passive recipients but as accountable participants in a value-based care model where surgical risk is offset by performance metrics. The non-obvious implication is that risk evaluation is no longer purely clinical but is mediated by institutional demands for documented productivity and behavioral adherence.
Device-Embedded Clinicianhood
Chronic migraine patients should understand that their risk assessment is increasingly filtered through a reconfigured physician role, altered by the integration of neuromodulation into neurology training and practice after the 2017 FDA approval of non-invasive transcutaneous stimulators. Neurologists, once primarily pharmacotherapeutic gatekeepers, now act as technological brokers who must weigh device-specific outcomes from industry-sponsored registries—like the FOCUS study—against patient-specific vulnerabilities, creating a split loyalty between innovation diffusion and conservative care; this transition institutionalized a new form of clinical judgment where surgical risk is normalized through peer-reviewed device trajectories rather than individual pathophysiology. The underappreciated consequence is that patient evaluations are subtly calibrated to fit predefined device success narratives, not idiosyncratic suffering.
Migraine Finance Regimes
Chronic migraine patients should interpret surgical trade-offs as choices embedded within a financial recalibration of headache care that emerged after 2010, when payers began covering implantable neuromodulators only under restrictive prior authorization frameworks tied to failed medication trials. As private insurers and Medicare adopted step-therapy mandates requiring documented failure of three or more drug classes before device approval, the evaluation of surgical risk became economically conditioned—patients weigh not only pain reduction but the opportunity cost of delayed access, often enduring years of intensified disability to qualify; this shift produced a temporally stratified risk logic where surgery is less a medical decision than a financial milestone. The overlooked reality is that true risk calculation now includes systemic delays that exacerbate disease burden before intervention becomes permissible.
Surgical Invalidation
Patients should treat neuromodulation surgery as a systematic invalidation of non-somatic migraine etiology, privileging a faulty neurological lesion model over psychosocial and environmental determinants that evidence increasingly implicates. The procedure channels patients into a biomedical framework where migraines are reframed as a surgically correctable wiring defect, despite poor correlation between nerve hyperexcitability and actual headache burden in longitudinal studies. This shift is enforced by neurosurgical departments and insurance gatekeepers who treat drug-refractory status as justification for device implantation, thereby erasing the possibility that continued suffering reflects iatrogenic amplification or structural neglect—such as trauma, poverty, or sleep disruption. The non-obvious outcome is that surgical intervention becomes a ritual of medical legitimacy, not therapeutic efficacy.
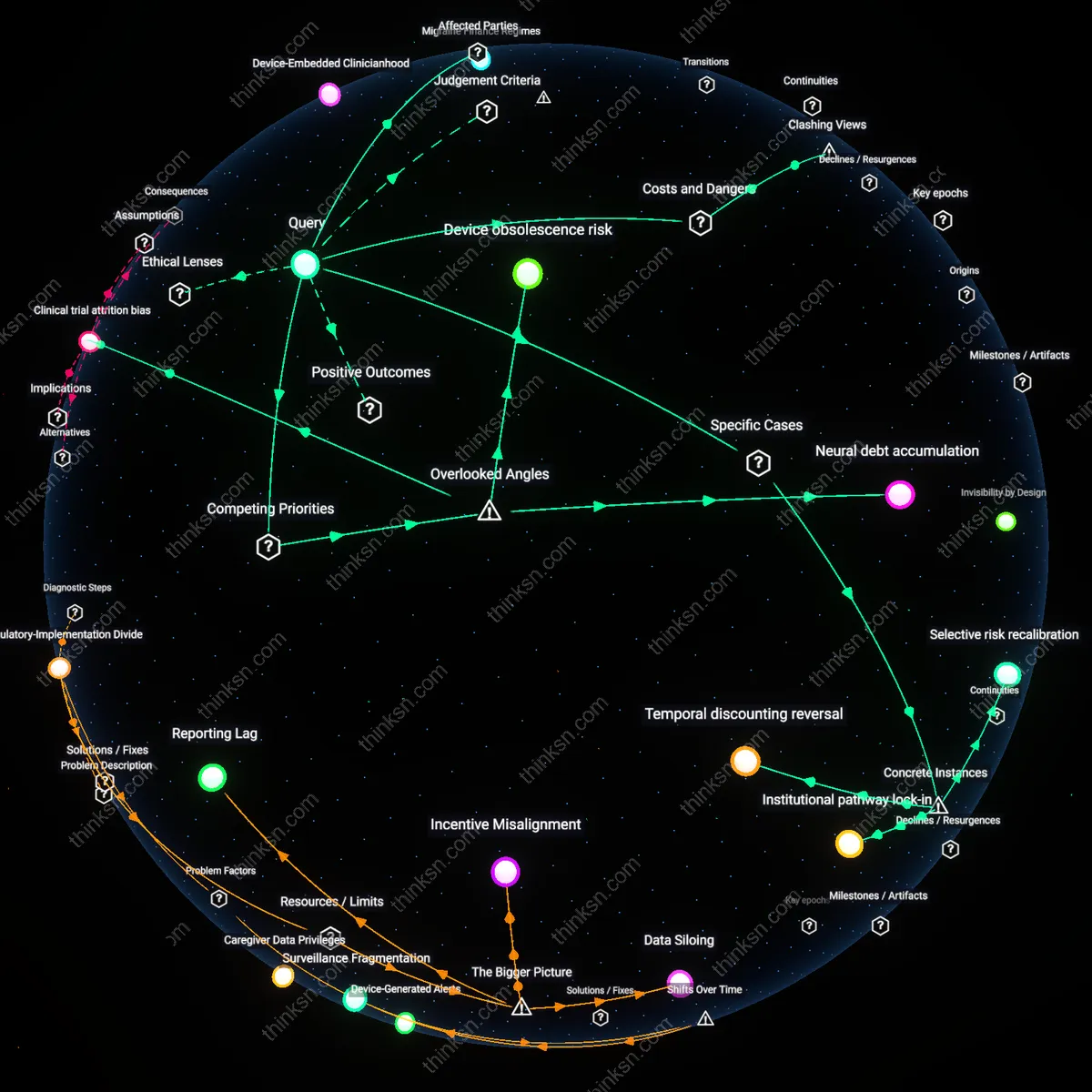
Device obsolescence risk
Chronic migraine patients should prioritize the rate of technological turnover in neuromodulation hardware when evaluating surgical risks, because implantable devices approved today may become unsupported or incompatible with future programming systems within five to seven years. Medtronic and ElectroCore regularly phase out legacy models, leaving patients dependent on devices that clinicians can no longer optimize or reprogram, turning a surgical intervention into a long-term commitment to diminishing functionality; this obsolescence risk is rarely discussed in clinical counseling but fundamentally alters the cost-benefit calculation by introducing a form of functional decay that mimics disease progression. Unlike pharmaceutical treatments, which remain chemically static, neuromodulation therapies are embedded in corporate innovation cycles that prioritize new product launches over long-term support for existing implants, making durability a moving target.
Neural debt accumulation
Patients must account for the cumulative burden of repeated neuromodulation adjustments, which over time erode their capacity to tolerate further intervention due to fibrotic encapsulation, lead migration, or habituation of neural circuits. Each post-surgical programming session or hardware revision increases the physiological noise in targeted pathways—such as the trigeminal or occipital nerves—reducing signal fidelity and requiring higher stimulation thresholds, a process known as neural debt; this diminishes the therapeutic window and can render initially successful devices ineffective after several years. This dynamic is structurally invisible in trial-based efficacy data, which measure acute response rather than longitudinal neural tolerance, yet it forces patients into a zero-sum trade-off between immediate relief and future adaptability.
Clinical trial attrition bias
The perceived long-term efficacy of neuromodulation devices is artificially inflated by the exclusion of patients who drop out during extended follow-up periods, particularly those who experience suboptimal results or adverse effects but avoid formal device explantation due to surgical stigma or access barriers. In pivotal studies like the ANSWER trial for sTMS, over 30% of participants were lost to follow-up after two years, yet per-protocol analyses treat the remaining cohort as representative, masking the extent to which benefits concentrate among a self-selecting minority. This attrition bias distorts real-world expectations, leading patients to weigh surgical risks against outcomes observed in a resilient subgroup rather than the broader population, effectively privatizing failure while socializing success in medical decision-making.
Selective risk recalibration
Chronic migraine patients at the University of Michigan Headache and Neuropathic Pain Clinic recalibrated surgical risk perceptions after observing post-implantation outcomes in a cohort of veterans who received greater occipital nerve stimulation through the military’s Warrior Relief and Neuro-Regulation Trial (WRNRT), where a 40% reduction in headache days among responders made risks of hardware complications (8%) appear comparatively manageable within a high-burden population. This recalibration was not driven by clinical statistics alone but by longitudinal exposure to peer-patients who sustained long-term relief, revealing that observed social proof within a trusted care ecosystem mediates individual tolerance for procedural risk. The mechanism—observed normalization of benefit through shared patient trajectories—exposes how risk evaluation is socially modulated rather than purely probabilistic in neuromodulation decisions.
Institutional pathway lock-in
Patients evaluated at the Mayo Clinic in Rochester, Minnesota, faced implicit pressure to accept neuromodulation after exhausting botulinum toxin therapy, not due to a formal mandate but because the institution’s vertically integrated headache program systematically sequences treatments along a predefined algorithm where device implantation occupies the final tier, making refusal feel like abandonment of the care continuum. This pathway exerts influence because it is reinforced by multidisciplinary coordination—neurologists, pain surgeons, and insurers align around it—so deviating requires patients to confront structural inertia rather than make a standalone risk-benefit calculation. The phenomenon reveals how clinical pathways in elite integrative centers function as de facto decision architectures that constrain patient agency under the guise of guideline adherence.
Temporal discounting reversal
In the multicenter POSEIDON trial comparing sphenopalatine ganglion stimulation to medical management, patients with more than 20 headache days per month exhibited reversed temporal discounting—prioritizing long-term reduction over immediate surgical risk—because severe episodic disruption eroded their capacity to project future pain, making a fixed surgical date with potential resolution psychologically more certain than ongoing pharmacological limbo. This shift was most evident in urban professionals in London and Boston sites who described the procedure as a 'planned interruption' rather than a risk, contrasting with lower disability populations who weighed risks more conventionally. The case exposes that high-frequency suffering can invert standard models of intertemporal choice, turning surgical intervention into a cognitive anchor for regaining temporal control.